 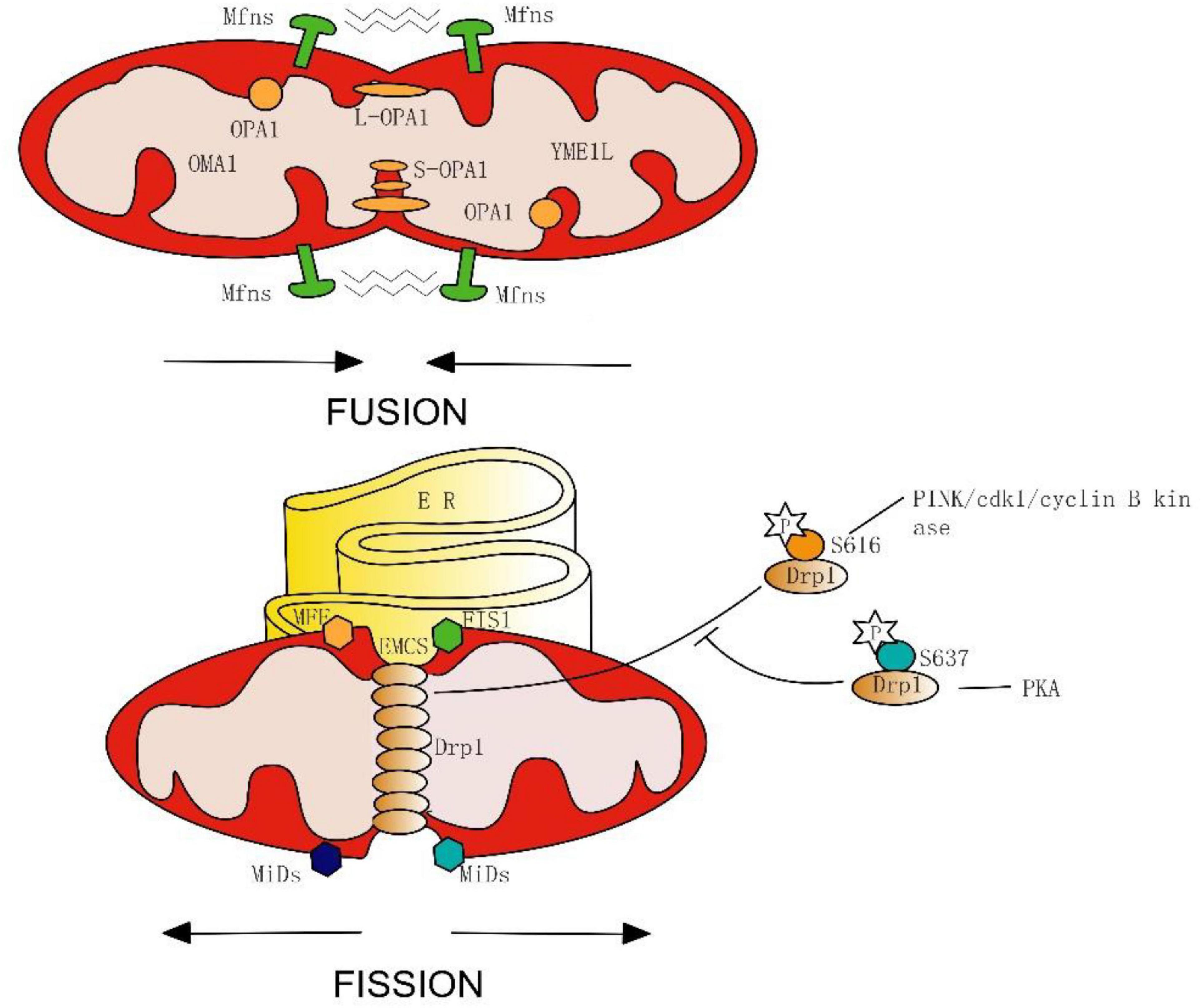
However, several observations suggest that this basic cellular process indeed plays a significant role in vertebrate cells. Doubts have been raised about the frequency and importance of mitochondrial fusion in cultured mammalian cells ( Enriquez et al., 2000). Both mitofusins are broadly expressed ( Rojo et al., 2002), and therefore their functional redundancy is unclear.ĭespite our increasing knowledge about the importance of mitochondrial fusion in lower eukaryotes, there is disagreement about its physiological role in vertebrates. Humans contain two Fzo homologues, termed mitofusin (Mfn)1 and Mfn2, that can alter mitochondrial morphology when overexpressed in cell lines ( Santel and Fuller, 2001 Rojo et al., 2002). Furthermore, there is a disruption of mating-induced mitochondrial fusion ( Hermann et al., 1998). fzo1Δ yeast form “petite” colonies that lack mitochondrial DNA (mtDNA) ( Hermann et al., 1998 Rapaport et al., 1998). In budding yeast, deletion of FZO1 disrupts the highly branched, tubular mitochondrial network typical of normal cells and results in numerous small spherical mitochondria. Disruption of Fzo prevents developmentally regulated mitochondrial fusion in postmeiotic spermatids and results in male sterility ( Hales and Fuller, 1997). * In Drosophila, Fzo is specifically and transiently expressed in spermatids. In both yeast and flies, mitochondrial fusion is controlled by the nuclearly encoded mitochondrial transmembrane GTPase, fuzzy onions (Fzo). Therefore, mitochondrial fusion is essential for embryonic development, and by enabling cooperation between mitochondria, has protective effects on the mitochondrial population. Strikingly, a subset of mitochondria in mutant cells lose membrane potential. We conclude that Mfn1 and Mfn2 have both redundant and distinct functions and act in three separate molecular complexes to promote mitochondrial fusion. Moreover, we find that Mfn1 and Mfn2 form homotypic and heterotypic complexes and show, by rescue of mutant cells, that the homotypic complexes are functional for fusion. Embryonic fibroblasts lacking Mfn1 or Mfn2 display distinct types of fragmented mitochondria, a phenotype we determine to be due to a severe reduction in mitochondrial fusion. However, whereas Mfn2 mutant embryos have a specific and severe disruption of the placental trophoblast giant cell layer, Mfn1-deficient giant cells are normal. We find that mice deficient in either Mfn1 or Mfn2 die in midgestation. The mitofusins, Mfn1 and Mfn2, have been shown to affect mitochondrial morphology when overexpressed. In this mini-review, we focus on plant mitochondrial fission and fusion, and compare them with mitochondrial fission and fusion in yeasts and animals, about which much has been learned in recent years.Mitochondrial morphology is determined by a dynamic equilibrium between organelle fusion and fission, but the significance of these processes in vertebrates is unknown. 
Using a novel photoconvertible fluorescent protein, we have recently shown that mitochondria in live plant cells rapidly undergo fusion and fission on a time scale of seconds. Little has been written about mitochondrial fusion in plants. On the other hand, Arabidopsis appears to have no genes similar to those involved in yeast mitochondrial fusion. These results show that DRP3A and DRP3B are functional homologues of the dynamin-related protein for mitochondrial fission in yeasts. Over-expression of DRP3A or DRP3B with point mutations caused severe elongation of mitochondria. DRP3A and DRP3B were localized to mitochondrial constricted sites and ends. The Arabidopsis genome has genes for two dynamin-related proteins, DRP3A and DRP3B, that are similar to genes involved in mitochondrial fission in yeasts. Single plant cells have hundreds of mitochondria that move around and change their shape through the processes of fission and fusion. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
